A Blog Series on Knowledge and Learning: 2
This blog introduces Legitimation Code Theory through the lens of Physical Chemistry education, drawing on the author's experience as a practitioner in the field. It is intended as an accessible entry point to the framework rather than an authoritative theoretical treatment. For the full scholarly framework, readers are directed to the work of Professor Karl Maton at the University of Sydney and the LCT Centre for Knowledge-Building: www.legitimationcodetheory.com
2.What is Legitimation Code Theory (LCT)?
Why Physical Chemistry feels so demanding, and what Legitimation Code Theory reveals about it
Have you ever wondered why Chemistry feels so different from studying literature or history? It is not just the formulae. There is something deeper going on, something about how knowledge itself is built, valued, and made accessible, or deliberately kept inaccessible, to students. This blog takes Physical Chemistry as its primary lens, the subdiscipline where the demands of vertical discourse are felt most acutely, while recognising that the argument it makes applies across Chemistry education more broadly. Drawing on Maton’s (2014) Legitimation Code Theory, it argues that the challenges students face are not simply a matter of ability or effort. They are structural, built into the way knowledge is organised, transmitted, and assessed. Understanding this is the first step toward changing it.
This blog draws on evidence from the United Kingdom and South African higher education contexts, two systems that together illuminate both the Global North and Global South dimensions of this problem. But the argument travels. The hidden rules of academic knowledge do not respect national borders.
Where LCT Comes From
LCT was developed by Australian sociologist Karl Maton, starting in the late 1990s and refined over the following decades (Maton, 2014). It did not emerge from nothing. Maton drew on two influential thinkers: Basil Bernstein, a British sociologist who explored how educational institutions organise and transmit knowledge (Bernstein, 1999), and Pierre Bourdieu, a French sociologist who examined how different fields operate and how cultural advantages are accumulated (Bourdieu, 1986). Maton took the best of both and turned them into something practical, a set of analytical tools that working educators and students can actually use, rather than purely abstract theory (Maton, 2014).
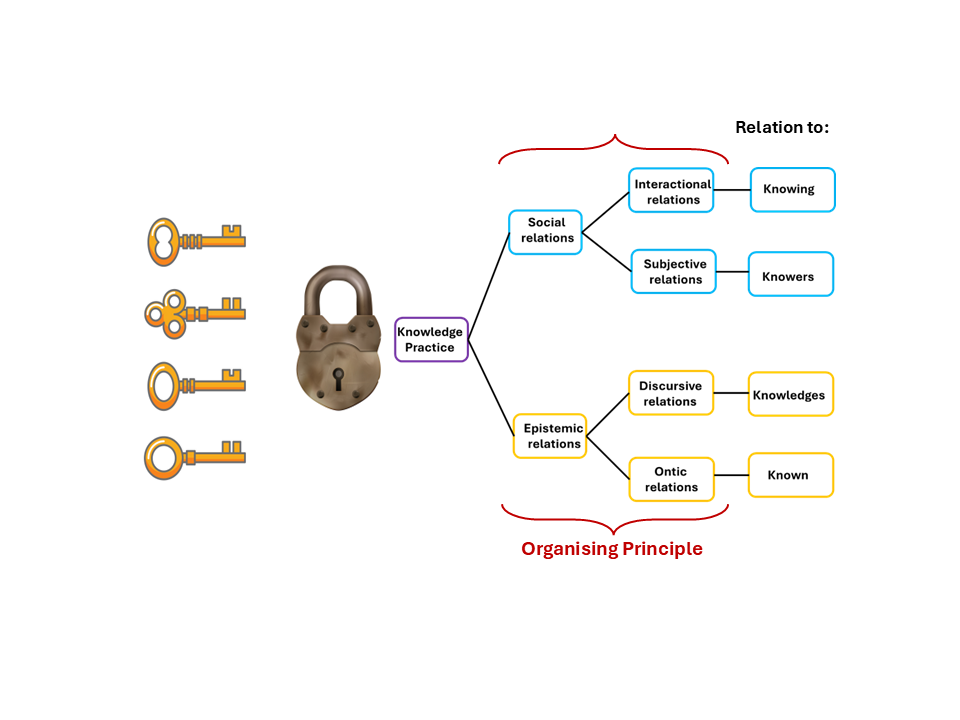
Figure 1 below helps make Maton’s analytical tools concrete. It is adapted from Maton (2014), and the lock and key imagery comes from Bernstein's insight that academic fields operate like locks, governed by hidden rules that determine who succeeds and why (Bernstein, 1999).
Think of it this way. Some students arrive at university already holding the keys. Their schooling, their home environment, the way they were taught to think and argue, all of it prepared them, often without them realising it, to play by the rules Chemistry demands. Other students arrive at the same door without those keys. Not because they are less capable, but because nobody ever handed them the keys in the first place.
The keys in Figure 1 represent the different organising principles of a field, the rules of the game. There is not one single key but several. What counts as legitimate knowledge in Chemistry operates on more than one level at once. The gold branches represent epistemic relations. These refer to what the knowledge is about, how abstract it is, and how it connects to the observable world. The blue branches represent social relations. These refer to who the knower is and the dispositions and habits of mind that the discipline expects them to bring. Together, these form what Karl Maton (2014) calls specialisation codes. These combinations shape every discipline and often determine, invisibly, who gains entry and who remains outside.
Figure 1. Keys, Locks and Legitimation: How LCT Organises Knowledge, adapted from Maton (2014) and Bernstein (1999).
What makes LCT particularly valuable is its practicality across many educational contexts. Chemistry education faces a well-known problem: students who performed well at A-level, Matric, or equivalent school-leaving qualifications frequently struggle once they arrive at university. LCT does not just describe this problem. It gives educators the tools to understand why it happens and where to intervene.
By analysing how knowledge shifts between concrete examples and highly condensed theoretical ideas, LCT helps educators identify where students lose track of meaning. In physical chemistry, for example, students often move rapidly from intuitive phenomena (such as diffusion, reaction rates, or energy changes) to abstract representations such as equations, mathematical models, and specialised terminology. LCT offers a framework for mapping these shifts and designing teaching that deliberately moves between real-world phenomena, conceptual explanations, and formal representations, helping students build stronger and more connected understanding. In this way, LCT helps make the structure of physical chemistry knowledge visible, allowing educators to design learning activities that guide students from everyday understanding to the abstract reasoning required in the discipline.
The Big Question: What Makes Knowledge 'Legitimate'?
LCT asks three questions that cut to the heart of how knowledge works in any discipline:
What counts as legitimate knowledge in a field?
What counts as a legitimate knower: what habits of mind does a discipline expect?
What are the legitimate practices: the accepted ways of thinking, reasoning, and communicating?
These questions matter in Chemistry. Is it enough to memorise reactions, or do you need to understand why they happen? Can you explain a concept in your own words, or must you use precise disciplinary language? LCT gives us the tools to ask who benefits when those expectations remain hidden.
Bernstein (1999) argued that university knowledge is “vertical”, it builds systematically, moves toward abstraction, and requires independent thinking. In Chemistry, this means moving beyond the periodic table or balancing equations, toward understanding why reactions behave as they do. Physical Chemistry makes this demand most sharply. Thermodynamics, quantum mechanics, and chemical kinetics require students to move fluidly between mathematical formalism, molecular-level reasoning, and macroscopic observation, building each concept on the last in a way that cannot be memorised or rehearsed.
Blackie (2014) demonstrated this directly, working with Chemistry students in South Africa, a context that mirrors widening participation challenges faced by universities globally. What students struggle with, she found, is often not the complexity of Chemistry but the degree of abstraction it demands. That distinction has direct implications for how we design curricula and assessments.
Yet the qualifications used to decide who enters university are well-documented as poor predictors of how students actually perform once they get there. Passing Chemistry at A-level or Matric often rewards memorisation and rehearsed technique rather than the deep conceptual thinking the subject demands. Knowing how to think scientifically, how to build an argument from evidence, and how to move between the symbolic, macroscopic, and sub-microscopic levels (which Johnstone (1982) identified as central to chemical understanding) is not equally available to all students. Those from under-resourced schools are frequently never taught these unwritten rules, regardless of which country they are in. What looks like a measure of ability may simply reflect how early a student was exposed to a particular, privileged way of knowing, what Bourdieu (1986) would call cultural capital. The real question is whether the Chemistry knowledge that gets a student into university is the same as the Chemistry knowledge that actually matters once they are there
How Knowledge is Organised: Vertical Discourse
When you enter university-level Chemistry, you are entering what Bernstein called vertical discourse, the structured, specialised knowledge of an academic discipline, very different from everyday ways of talking and thinking (Bernstein, 1999). In vertical discourse, knowledge does not simply accumulate; it transforms. Each new concept reorganises what you already knew. Understanding bonding changes how you see atomic structure. Understanding thermodynamics reframes how you think about reactions. New knowledge does not sit beside the old; it integrates with it (Maton, 2014; Bernstein, 1999).
What LCT adds to Bernstein’s insight is a practical question: are these transformative demands made explicit to students, or are they left to figure them out alone? Chinaka (2021), studying first-year Chemistry students at a South African university, found that when teaching the second law of thermodynamics, students were frequently left stranded in abstraction, unable to connect theoretical principles back to observable chemical phenomena. The problem is not the knowledge itself. It is the assumption that students will navigate its demands without guidance. This assumption is both widespread and costly.
Boughey and McKenna (2021) deepen this point. Examining South African higher education, a system whose challenges reflect in concentrated form, the equity problems facing universities globally. They argue that institutions consistently treat access to powerful disciplinary knowledge as though it were equal, when the evidence shows it is not. Students are positioned as decontextualised learners, as though their backgrounds, schooling, and social circumstances are irrelevant to how they engage with Chemistry. The result is a university that presents itself as a meritocracy while quietly favouring those who already speak its language. Vertical discourse, in this reading, is not just a feature of Chemistry. It is a mechanism through which inequality is reproduced, unless educators actively work to make its demands visible and navigable for all students.
Knowledge Structures: Why Chemistry Thinks the Way It Does
Bernstein drew a crucial distinction between hierarchical and horizontal knowledge structures (Bernstein, 1999; Maton & Muller, 2007). In Chemistry, knowledge is hierarchical. Concepts such as atomic structure, chemical bonding, thermodynamics, and reaction kinetics do not sit in isolation. They build on each other and integrate into broader explanatory frameworks. Discoveries extend or refine existing theory rather than simply accumulating alongside it (Maton, 2006; 2014).
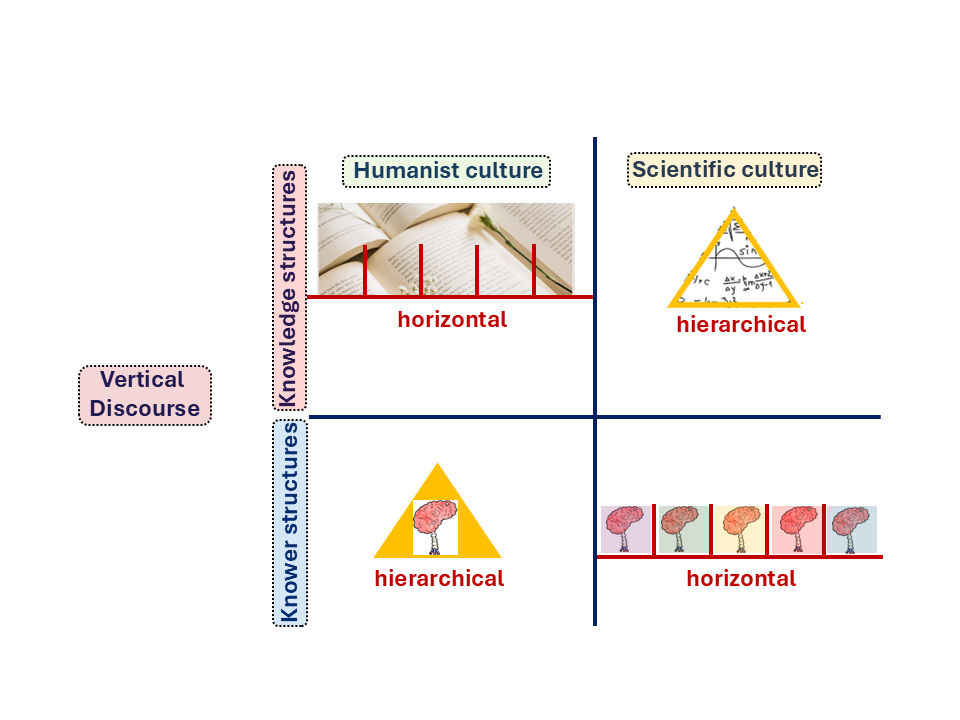
Figure 2. Knowledge structures and knower structures across humanist and scientific cultures within vertical discourse, adapted from Maton (2006; 2014)
Figure 2 shows how this plays out across humanist and scientific cultures. Chemistry sits firmly in the scientific culture quadrant, characterised by hierarchical knowledge structures, yet comparatively horizontal knower structures. In Chemistry, it is what you know, not who you are, that determines legitimate participation in the field.
This contrasts with fields such as literary studies or sociology, where multiple theoretical perspectives can coexist without being integrated into one overarching theory, what Bernstein called horizontal knowledge structures (Bernstein, 1999). Students notice this difference instinctively. In Chemistry, you cannot skip foundations. You need atomic theory before bonding, bonding before thermodynamics. The hierarchy is real and unforgiving.
What is particularly troubling is that this hierarchical demand is not always reflected in the materials used to teach Chemistry. Wei et al. (2025), analysing secondary school chemistry textbooks through LCT’s concepts of semantic gravity and semantic density, found that the overwhelming majority of knowledge in those textbooks favoured concrete, context-dependent content over abstract, complex understanding. Their analysis suggests textbooks avoid precisely the kind of abstraction that university Chemistry demands. Students are therefore arriving at university unprepared for vertical, hierarchical thinking, not because they lack ability, but because the very materials designed to teach them have not asked it of them. That is a structural problem, and it shows up across educational systems worldwide.
Cultural Capital and the Hidden Rules of Chemistry
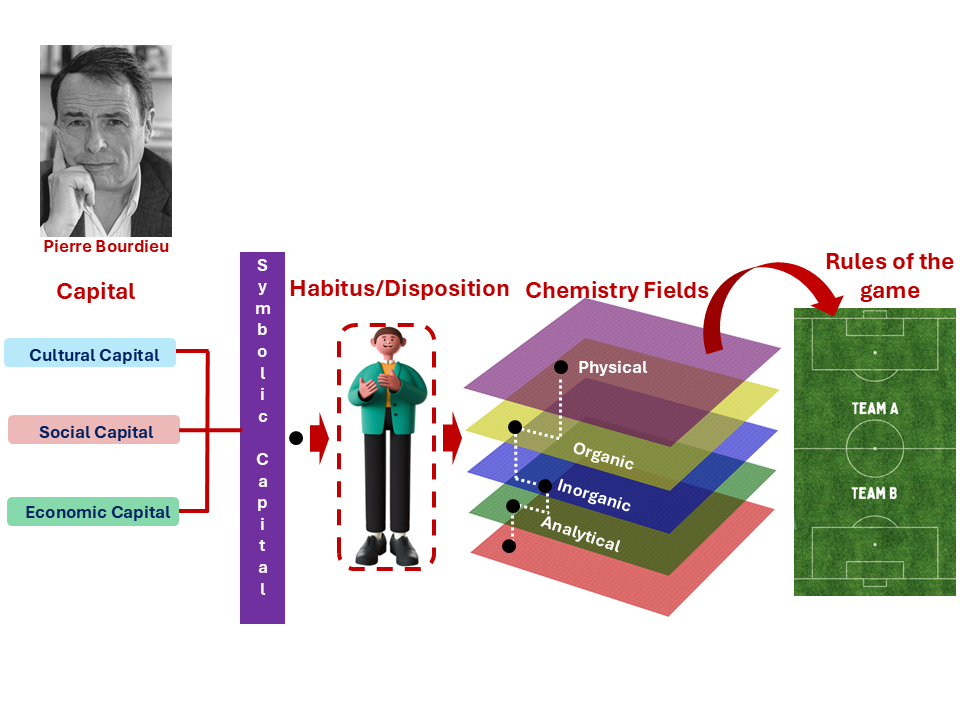
Figure 3: The relationship between capital, habitus, and field in Chemistry education. Adapted from Bourdieu (1986) and Bourdieu and Passeron (1977).
Picture of Pierre Bourdieu: Lambert, B. (1996). Pierre Bourdieu. Wikimedia Commons. Available at: https://commons.wikimedia.org/wiki/File:Pierre_Bourdieu_(1).jpg (Accessed: 12 March 2026).
Figure 3 shows how the three forms of capital, cultural, social and economic. They combine to shape the dispositions and habits of mind that students bring into Chemistry, ultimately determining whether they recognise and can play by the unwritten rules of the game that the discipline demands. In Chemistry, cultural capital shows up in subtle but powerful ways. Students who arrive already comfortable with mathematical reasoning, scientific notation, or the logic of experimental design have a head start, not because they are more capable, but because they already speak some of the language (Maton, 2014; Bourdieu & Passeron, 1977).
This is where LCT goes beyond Bourdieu. Bourdieu shows us that cultural capital is unevenly distributed. LCT gives educators the tools to actually do something about it. Making the rules of the game explicit, such as what counts as a good explanation, the level of precision expected, and how to construct a valid argument, can help students better understand what is required of them. By clarifying these expectations, educators can help level the playing field and support more students to succeed (Maton, 2014; Blackie, 2014). Deep educational inequality is not an abstract concern. It is the daily reality for students navigating the schooling disparities of South Africa, the widening participation agenda of the UK, or the challenges of being the first in their family to attend university anywhere in the world. Whether Chemistry remains the preserve of the already privileged, or becomes genuinely accessible to all who have the capacity to engage with it, depends in no small part on whether educators are willing to make those rules visible.
Concluding Remark
LCT does not just describe why Chemistry feels demanding and exclusionary. It points toward a solution. If the hidden rules of academic knowledge can be identified, they can be taught. That is not a small claim. It is an act of educational justice.
However, the framework is not without its limits. Georgiou, Maton, and Sharma (2014) identified what they call the Icarus Effect: the risk that students either stay too close to concrete examples and never reach the abstraction that Chemistry demands, or fly so deeply into abstraction that they lose touch with the observable world entirely. In Physical Chemistry, where thermodynamics, quantum mechanics, and kinetics demand constant movement between the abstract and the concrete, this is not a theoretical risk. It is the everyday experience of many struggling students.
LCT itself invites an important question: Is the framework advocating for vertical knowledge, or simply exposing it? The answer matters. Its real strength lies not in endorsing academic hierarchies but in making them visible enough to be navigated by everyone, not just those who inherited the map. As Boughey and McKenna (2021) remind us, universities present themselves as meritocracies while quietly favouring those who already speak the language. Chemistry is no exception. Identifying that problem is the first step. The harder work of changing how Chemistry is actually taught remains ongoing. A framework is only as powerful as its application, and that rests with the educators and students willing to engage with it seriously, wherever they are in the world.
Coming Next
The Epistemic Plane: Understanding How Chemistry Positions Its Learners
Legitimation Code Theory does not stop at knowledge structures. It also asks what counts as legitimate knowledge in a discipline. Karl Maton (2014) directs attention to the epistemic plane. This concerns the relationship between knowledge, its object, and the knower. In Chemistry, this raises an uncomfortable but necessary question. Does the discipline foreground what students know while paying less attention to who they are as learners and what they bring with them? This question, and its practical implications for teaching Chemistry in different contexts, will be explored in the next blog in this series.
References
Bernstein, B. (1999). Vertical and horizontal discourse: An essay. British Journal of Sociology of Education, 20(2), 157-173.
Blackie, M.A.L. (2014). Creating semantic waves: Using Legitimation Code Theory as a tool to aid the teaching of chemistry. Chemistry Education Research and Practice, 15(4), 462-469.
Bourdieu, P. (1986). The forms of capital. In J. Richardson (Ed.), Handbook of theory and research for the sociology of education (pp. 241–258). Greenwood Press.
(https://www.marxists.org/reference/subject/philosophy/works/fr/bourdieu-forms-capital.htm)
Bourdieu, P., & Passeron, J.C. (1977). Reproduction in education, society and culture. Sage.
Boughey, C., & McKenna, S. (2021). Understanding higher education: Alternative perspectives. African Minds.
Chinaka, T.W. (2021). Introducing the second law of thermodynamics using Legitimation Code Theory among first year chemistry students. Cypriot Journal of Educational Sciences, 16(3), 981-994.
Georgiou, H., Maton, K., & Sharma, M. (2014). Recovering knowledge for science education research: Exploring the “Icarus Effect” in student work. Canadian Journal of Science, Mathematics and Technology Education, 14(3), 252–268.
Johnstone, A.H. (1982). Macro and microchemistry. School Science Review, 64(227), 377-379.
Maton, K. (2006). On knowledge structures and knower structures. In R. Moore, M. Arnot, J. Beck & H. Daniels (Eds.), Knowledge, power and educational reform. Routledge.
Maton, K. (2014). Knowledge and knowers: Towards a realist sociology of education. Routledge.
Maton, K. & Muller, J. (2007) A sociology for the transmission of knowledges, in Christie, F. & Martin, J. (eds) Language, Knowledge and Pedagogy: Functional linguistic and sociological perspectives (pp. 14-33). London: Continuum.
Wei, B., Zhan, Z., Jiang, Z. & Yu, L. (2025). Representation of learning outcomes stipulated by the intended curriculum in four series of chemistry textbooks: Based on Legitimation Code Theory. Research in Science Education, 55(2), 445-462