A Blog Series on Knowledge and Learning: 4
This blog introduces Legitimation Code Theory through the lens of Physical Chemistry education, drawing on the author's experience as a practitioner in the field. It is intended as an accessible entry point to the framework rather than an authoritative theoretical treatment. For the full scholarly framework, readers are directed to the work of Professor Karl Maton at the University of Sydney and the LCT Centre for Knowledge-Building: www.legitimationcodetheory.com
4. The Social Plane: Who Gets to Belong in Physical Chemistry?
Physical Chemistry doesn't ask who you are. It just assumes you were already prepared.
In the previous blog 3, we spent all our time on the gold branches in Figure 1 from Blog 2. We examined epistemic relations, the Epistemic Plane, and the four ways a student can be positioned in relation to the knowledge of Physical Chemistry. We ended with Tom, who earned an A on his atomic structure course and left with a broken mental model of the atom. The gap between his grade and his understanding was a gap in how he was positioned in relation to the knowledge.
But there is a second set of branches in Figure 1, below. The blue one raises a different and more uncomfortable question. Not what kind of knowing does Physical Chemistry require, but who does Physical Chemistry quietly expect its students to be? That is what this blog is about.
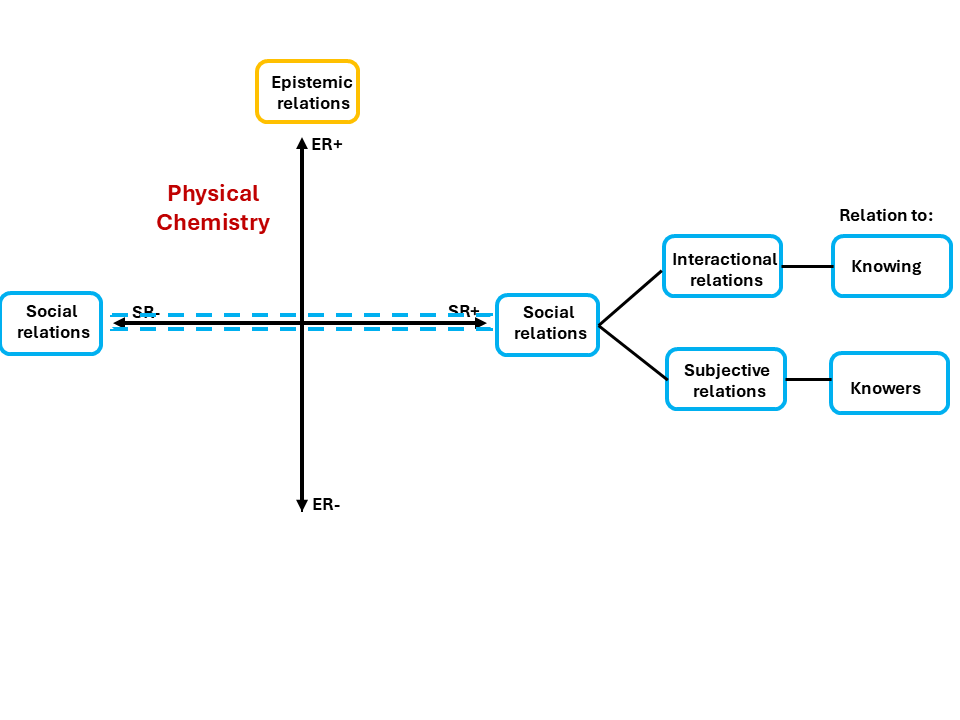
Figure 1: Specialisation plane focusing on the Social Relation, adapted from Maton(2014)
What the Blue Branches Open Up
Recall from the previous blog that the Specialisation Plane has two axes. Epistemic relations sit on the vertical axis, measuring how strongly the knowledge is bound. Social relations sit on the horizontal axis, measuring how much the knower matters. Physical Chemistry sits high on the vertical axis and to the left. Strong epistemic relations, weaker social relations. What you know is everything. Who you are is downplayed.
That is Physical Chemistry's formal position. But as Figure 1 shows, social relations are not one thing. They branch further. When these branches are examined, the discipline’s claim to neutrality about the knower becomes harder to sustain.
LCT breaks social relations into two independent dimensions. Subjective relations (SubR) ask whether legitimacy depends on who you are, your identity, your background, or your social category. Does being from a particular group, defined by gender, race, class, language, or schooling, make you more or less likely to be recognised as someone who belongs? Interactional relations (IR) ask whether legitimacy depends on how you came to know, through what practices, mentoring, and sustained immersion, your ways of engaging with knowledge were built. Did you develop the habits of mind that Physical Chemistry quietly requires before you walked through the door?
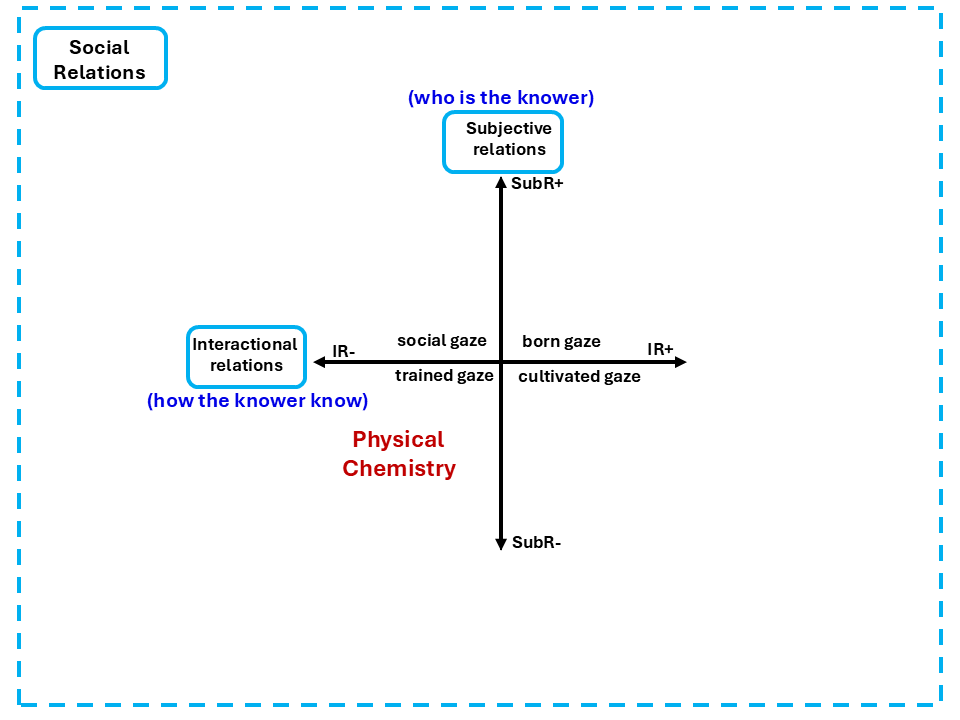
Both can vary independently. Together, they generate the Social Plane, the four quadrants shown in Figure 2.
Figure 2: The Social Plane — Subjective relations (SubR) and Interactional relations (IR) axes,with the four gazes: social gaze, born gaze, cultivated gaze, trained gaze. Adapted from Maton (2014).
Where Physical Chemistry Says It Sits
Physical Chemistry's formal position on the Social Plane is the trained gaze, bottom left, where both subjective and interactional relations are weak. It does not matter who you are. It does not matter how you came to develop your understanding. You master the knowledge, you belong. Your identity is irrelevant and your educational history is irrelevant. Produce the right answer, and you are a legitimate participant in the discipline.
This is a genuine commitment, and it matters. It is what makes Physical Chemistry, like physics and mathematics, different from disciplines where the identity of the knower is explicitly part of what is being valued. In fine art, in music performance, in creative writing, your particular self is part of the product. In Physical Chemistry, in principle, it is not. The trained gaze is the discipline's promise that it is open to anyone.
But here is the problem. A promise can be sincere and still operate in ways that contradict it in practice.
This creates a tension that is easy to miss. Formally, Physical Chemistry operates as a knowledge code. Legitimacy depends only on mastery of the knowledge. But in practice, success often depends on prior ways of thinking, learning, and engaging with that knowledge. Ways that students were expected to have developed before they arrived. The discipline presents itself as operating with a trained gaze, while quietly relying on elements of a cultivated gaze. Both are present at the same time. But only one is made visible.
This is not a matter of the discipline being insincere. The commitment to knowledge over knower is real. The issue is that the conditions required to access that knowledge are not made explicit. What appears to be neutrality at the level of principle becomes selective in practice, because the prior formation the discipline assumes is unevenly distributed. Think of it this way: the discipline, as a structure, can enable learning. But it only does so when a particular mechanism is already in place, the cultivation of certain ways of thinking and engaging with knowledge before university entry (Archer, 2000). Where that has not happened, the structure alone is not enough.
The Gap Between the Promise and the Practice
Lindstrøm and Sharma (2011) tracked the physics self-efficacy of first-year students at the University of Sydney across four measurement points throughout the year. Self-efficacy is simply a student's belief in their ability to succeed, solve problems and handle difficulties, which matters a great deal. Students with low self-efficacy treat hard problems as threats rather than challenges.
The findings were striking. Males consistently reported higher self-efficacy than females. That gap was there at entry, before a single university lecture had been given, and it persisted all year. Females with A grades reported similar self-efficacy to males with C grades. The grade was not protecting the student's belief in their own ability. Something else was operating. Something the knowledge itself and the assessment of it could not fix. Prior formal instruction in physics also mattered. Students who had not studied physics at school entered with lower confidence, and that disadvantage tracked right through the year. The knowledge code says none of this should matter. The data says it does.
Self-efficacy is not itself a social relation in LCT terms. It is an outcome of what we observe when social relations have already done their work on a student before they arrive. But it is a useful signal. The gender gap points toward subjective relations: who you are perceived to be, and whether people like you are routinely seen as legitimate participants in physics. The prior instruction gap points toward interactional relations: what practices you were inducted into, whether you encountered physics as something you reason about or merely something you execute. Both are operating here, and crucially, neither is visible in a grade.
The picture from chemistry calculations tells the same story from a different angle. Rusek et al. (2026) studied 547 first-year chemistry students across six universities. They assessed five core calculation areas: molar concentration, mass fraction, stoichiometry, dilution, and pH. All standard secondary school curriculum. All are formally required at university entry. The mean score across the cohort was 39.5%. More than half the students scored below 50%. Only a small fraction reached 70% or above.
What makes this particularly striking is what did not predict the outcomes. Students from chemistry-specialist technical schools, who had received significantly more instructional time in chemistry and extensive laboratory training, did not consistently outperform students from general academic schools. Students in chemistry teacher programmes performed at virtually the same level as students in professional chemistry programmes. More time in the system. More hours on the topic. A more specialised track. None of it reliably moved the needle. The knowledge code says mastery is what counts. The data says something else is determining who arrives prepared.
What Lindstrøm and Sharma (2011) measured was self-efficacy, not LCT's social relations directly, but the two are connected through cause and effect. The gender gap in self-efficacy reflects the accumulated effect of subjective relations: the discipline's historical identity as a masculine domain shapes who feels they belong in it, and that shaping happens long before university. The prior instruction gap reflects the accumulated effect of interactional relations: students who were inducted into physics as a reasoning practice rather than a content body to be memorised arrive differently equipped, not because they are smarter, but because they were formed differently. Both arrive in the room with the student. The outcomes we observe (lower confidence, uneven mastery, grades that do not reflect understanding) are not random. They are produced by structures and mechanisms that were operating long before anyone walked into a university lecture (Archer, 2000).
When a Trained Gaze Depends on Cultivation
Ellery (2021), studying a university science access programme in South Africa, found something that makes the mechanism visible. Most science courses do not operate with only one code. They operate with two, simultaneously. The first is the knowledge code, explicitly stated, assessed, and rewarded. Master the content, and you pass. The second is a knower code that is never stated at all. It operates through the pace of the course, the volume of material, the assumption that students will regulate their own understanding independently, and the expectation that they will engage at depth without being told to. This second code quietly sorts students. Those whose prior schooling already produced those habits of mind pass through. Those whose schooling did not are filtered, without anyone naming the filter.
It is like a course that says “we will teach you to swim” but begins in the deep end, assuming you are already comfortable in the water.
In LCT terms, what Ellery describes is code layering. A knowledge code operates explicitly at the surface: this is what is taught, assessed, and rewarded. But a latent knower code operates beneath it, privileging students who have already developed particular interactional formations (the problem-solving routines, the modelling practices, the disciplinary discourse) before they arrived. The trained gaze is what the discipline claims. The cultivated gaze is what it quietly relies on. Both codes are present, and only one is named.
The cultivated gaze, upper right in the Social Plane in Figure 2, legitimates knowers not through who they are, but through how they came to know. Through sustained immersion in particular practices, models, and ways of engaging with knowledge. Students who enter Physical Chemistry having been shaped by cognitively rich schooling, who learned early to regulate their own understanding, who encountered physics and chemistry as a practice rather than just a body of content, have already been cultivated. Students who have not been cultivated are not deficient. They simply have not yet had access to the cultivation that the discipline quietly assumes they already have.
Sánchez Gómez and Suárez (2025) put this in sharp relief. They argue that a chemistry identity is not built through content knowledge alone. It requires three things. First, competence in the representational practices of chemistry, including the ability to use molecular graphs, reaction mechanisms, and chemical equations to draw inferences. Second, performance, which involves actively engaging in these practices in social settings. Third, recognition, which includes being seen by others in the discipline, and seeing yourself, as someone who does chemistry. They call this the Trinity Model. The point for this blog is direct: a student who has learned to execute procedures but has never been drawn into the social and representational practices of the discipline, who has never had their reasoning recognised by an expert as chemistry reasoning, has not yet developed a chemistry identity in any meaningful sense. They are executing, but they are not yet belonging. That gap is not a matter of intelligence. It is a matter of access to the practices that constitute the discipline.
Boughey and McKenna (2016) name the consequence clearly, as the decontextualised learner. A student whose difficulties are attributed to their own shortcomings, their attitude, their effort, their ability, when the actual source of the difficulty is structural. The dominant model in universities treats language and ways of thinking as neutral skills that students either have or lack. It does not ask where those skills come from, who had access to them, and who was never offered them. It does not see the barrier because the barrier was never named. Sánchez Gómez and Suárez (2025) add a further layer, that the barrier is also institutional. University degrees are designed around a single, well-defined disciplinary identity. The curriculum, the assessments, and the pace of a chemistry or physics degree are built to induct students into one specific community of practice. Any student who arrives without the prior formation that community quietly assumes faces not just a gap in preparation, but a structural mismatch between who the institution expects them to be and who they currently are.
The Language Problem Nobody Talks About
The language of Physical Chemistry is a case study in this. Physical Chemistry does not just use specialist vocabulary. It uses words that appear in everyday language but mean something entirely different in a chemistry context. Spontaneous in everyday life means impulsive or unplanned. In thermodynamics it means a process that occurs without external input, given the right conditions. Work in everyday life means effort. In thermodynamics it is a precise form of energy transfer. Strong in everyday life is a compliment. In chemistry it refers to the degree of dissociation of an acid. Free in everyday life suggests unrestricted. In Gibbs free energy it means available to do useful work.
Quílez (2019) documents this systematically. These are not accidental overlaps. They are structural sources of student difficulty, arising from the history of how scientific concepts developed and were named. And they are compounded by the fact that Physical Chemistry uses technical terms whose meanings have shifted across different historical traditions and textbooks. Students can encounter the same word with different meanings in different contexts. Entropy has five competing metaphors with no scientific consensus. The language of electrochemistry carries specific meanings across quantum mechanics, classical thermodynamics, and everyday description simultaneously.
A student from a schooling background where these terminological traps were explicitly named, where the teacher said “when I use the word spontaneous in chemistry, here is what I mean and here is why it differs from everyday usage,” has a navigational advantage that has nothing to do with intelligence or effort. A student for whom this was never made explicit faces a barrier that the discipline does not acknowledge. Science teachers, as Quílez (2019) notes, rarely see themselves as language teachers. The result is that the linguistic demands of Physical Chemistry function as an invisible gate. Wide open for students who arrived already carrying the key, and invisible to those who were never told it existed.
This is the cultivated gaze in operation. Not through explicit preference for certain identities. Through the accumulated effect of practices, norms, and expectations that were never made explicit, and that differentially reward the students who had already been formed in the right way.
Rusek et al. (2026) reveal the same mechanism from a different angle. Calculation performance was weak and uniform across all school types and study programmes. Why? Because students had been taught calculations as isolated procedural topics, typically concentrated in one block of upper-secondary school and rarely revisited. The result was a cohort that could execute familiar algorithms under familiar conditions. But struggled the moment the same concept appeared in a slightly different form. The discipline assumed students would arrive with quantitative reasoning integrated into their chemical understanding. What had actually been cultivated was procedural execution, disconnected from conceptual meaning. The gap between what was assumed and what had been built was invisible to both the discipline and the students.
What This Means in Practice
So what do we do with this? The Social Plane is not just a diagnostic tool. It points toward something concrete.
The discipline formally promises a trained gaze, legitimacy through knowledge, not through the knower. Honouring that promise means making the implicit cultivated gaze visible and teachable. There are two directions.
The first is about subjective relations, the who. The gender gap in physics self-efficacy was present from entry and persisted through the year regardless of grade (Lindstrøm & Sharma, 2011). A better assessment will not fix this. It requires explicit attention to who is seen in the room, who is heard in discussions, and who is affirmed when they make progress. A sense of belonging correlates with self-efficacy, and self-efficacy is one of the strongest predictors of persistence. Students who do not see people like themselves succeeding in physical science are less likely to develop the confident relationship with the knowledge the discipline requires. Making expert thinking visible, showing students that the reasoning they are developing is physics reasoning, that their questions are physics questions, is not a soft add-on. It is directly relevant to whether the knowledge code's promise holds.
The second is about interactional relations, the how. The cultivated gaze becomes teachable when you stop assuming students arrived with the practices the discipline requires and start building those practices explicitly into the course. This means narrating expert reasoning out loud rather than demonstrating only the finished product. It means teaching the language of Physical Chemistry as an explicit object of instruction, naming the terminological traps, explaining what spontaneous means here and why it differs from everyday usage. It means treating calculations not as isolated procedural exercises covered once and assumed mastered, but as tools for reasoning about chemical phenomena that need to be revisited, integrated, and made meaningful. It means creating opportunities for students to actually use the representational practices of the discipline in social settings, drawing a mechanism and explaining it, not just getting it right on an exam. Sánchez Gómez and Suárez (2025) are clear that recognition, being seen as someone who reasons chemically, only comes through performance, and performance only comes through practice that is embedded in the social context of the discipline. You cannot develop a chemistry identity in isolation from the community that confers it. Rusek et al. (2026) are clear: improving performance in chemical calculations is unlikely to come from more practice when that practice emphasises routine execution. What is needed is instruction that connects numerical results to chemical meaning, and that makes the reasoning visible rather than just the answer. Ellery (2021) reaches the same conclusion from a different direction: without access to both the knowledge code and the implicit knower code, students are unlikely to succeed, the two are not separable.
Concluding Remark
Physical Chemistry requires a cultivated gaze but teaches as if a trained gaze were sufficient. That gap is not a matter of poor teaching or student deficit. It is a structural feature of how the discipline misrecognises its own conditions of access. The knowledge code is real, and the commitment to openness is real. But the prior formation that makes the knowledge code accessible is not equally distributed, not made explicit, and not taught, and so the promise of meritocracy quietly produces the opposite.
Physical Chemistry's formal commitment to the trained gaze is worth taking seriously. It is one of the discipline's genuine strengths. But a gap between the formal promise and the lived reality of students is not honoured by insisting the promise is true. It is honoured by closing the gap.
The Social Plane tells us where to look. The question is not why these students don’t belong. The right question is: what has the discipline assumed they already were before they arrived, and how can we make those assumptions into explicit practices that everyone can access?
Physical Chemistry does not need to lower its standards to address this. Purist insight, the integrated physical-mathematical understanding we explored in the previous blog, is still the goal. What changes is the assumption that students will reach it by absorbing an implicit, cultivated gaze they were never taught. The issue is not that Physical Chemistry values knowledge over knower. It is that access to that knowledge is shaped by forms of prior cultivation that the discipline does not name. Making those forms visible and teachable is not a concession. It is what the trained gaze actually requires.
This matters most in contexts where prior cultivation has been unevenly distributed. In systems such as South Africa, where students arrive with highly varied educational experiences, the assumption that everyone has already developed the ways of thinking, reasoning, and engaging that Physical Chemistry demands simply does not hold. The outcomes we observe, uneven confidence, lower persistence, grades that do not translate into understanding, are not personal failings. They emerge from the interaction between the structural assumptions of the discipline and the uneven reach of the mechanisms that were supposed to cultivate those assumptions before university (Archer, 2000). Where that cultivation has not reached, more students are left navigating expectations that were never made visible. Closing that gap is not a welfare concern separate from academic standards, it is the academic work.
Coming Next
Semantic Waves and Conceptual Meaning in Physical Chemistry
The Epistemic Plane told us what kind of knowing Physical Chemistry requires. The Social Plane told us who the discipline quietly assumes its learners already are. The next dimension, Semantics, explores how conceptual meaning is structured and how it moves in Physical Chemistry. How context-dependent is the knowledge? How condensed? And what happens when teaching keeps meaning at one level rather than moving deliberately between the concrete and the abstract? That is the focus of the next blog.
References
Archer, M.S. (2000). Being human: The problem of agency. Cambridge University Press.
Boughey, C. & McKenna, S. (2016). Academic literacy and the decontextualised learner. CRISTAL: Critical Studies in Teaching and Learning, 4(2), 1–9.
Ellery, K. (2021). Widening access in science: Developing both knowledge and knowers. In K. Maton, J.R. Martin & Y.J. Doran (Eds.), Teaching Science: Knowledge, Language, Pedagogy (pp. 187–204). Routledge.
Lindstrøm, C. & Sharma, M.D. (2011). Self-efficacy of first-year university physics students: Do gender and prior formal instruction in physics matter? International Journal of Innovation in Science and Mathematics Education, 19(2), 1–19.
Maton, K. (2014). Knowledge and knowers: Towards a realist sociology of education. Routledge.
Quílez, J. (2019). A categorisation of the terminological sources of student difficulties when learning chemistry. Studies in Science Education, 55(2), 121–167.
Rusek, M., Koperová, D., Bártová, I., Machková, V., Chytrý, V., Sirotek, V., Štrofová, J. & Šmejkal, P. (2026). Challenges in chemistry calculations at university entry: Evidence from a multi-institutional study. Journal of Chemical Education. https://doi.org/10.1021/acs.jchemed.6c00022
Sánchez Gómez, P.J. & Suárez, M. (2025). Plurality and identity: on the educational relations between chemistry and physics. Chemistry Education Research and Practice, 26, 53–64.