A Blog Series on Knowledge and Learning: 3
This blog introduces Legitimation Code Theory through the lens of Physical Chemistry education, drawing on the author's experience as a practitioner in the field. It is intended as an accessible entry point to the framework rather than an authoritative theoretical treatment. For the full scholarly framework, readers are directed to the work of Professor Karl Maton at the University of Sydney and the LCT Centre for Knowledge-Building: www.legitimationcodetheory.com
3. The Epistemic Plane: Understanding How Chemistry Positions Its Learners
Why passing Physical Chemistry and understanding Physical Chemistry are not always the same thing, and what LCT reveals about the difference
In the previous blog 2, we introduced the keys and locks image to make a fundamental point about how knowledge works in Physical Chemistry. Recall that the gold branches in that figure represent epistemic relations: what the knowledge is about, how abstract it is, and how it connects to the observable world. The blue branches represent social relations: who the knower is, and the habits of mind and dispositions the discipline expects them to bring. Maton (2014) describes these as specialisation codes, underlying patterns in a discipline that determine how knowledge and learners are valued.
Physical Chemistry sits in what that framework calls a knowledge code. What you know is the primary currency. Who you are matters far less. That is why the subject can feel, on the surface, like a meritocracy (produce the right answer, and you pass). This blog centres on an uncomfortable truth. The knowledge that gets a student through a chemistry exam is not always the same as the knowledge Physical Chemistry actually requires. Figure 1 helps us see why.
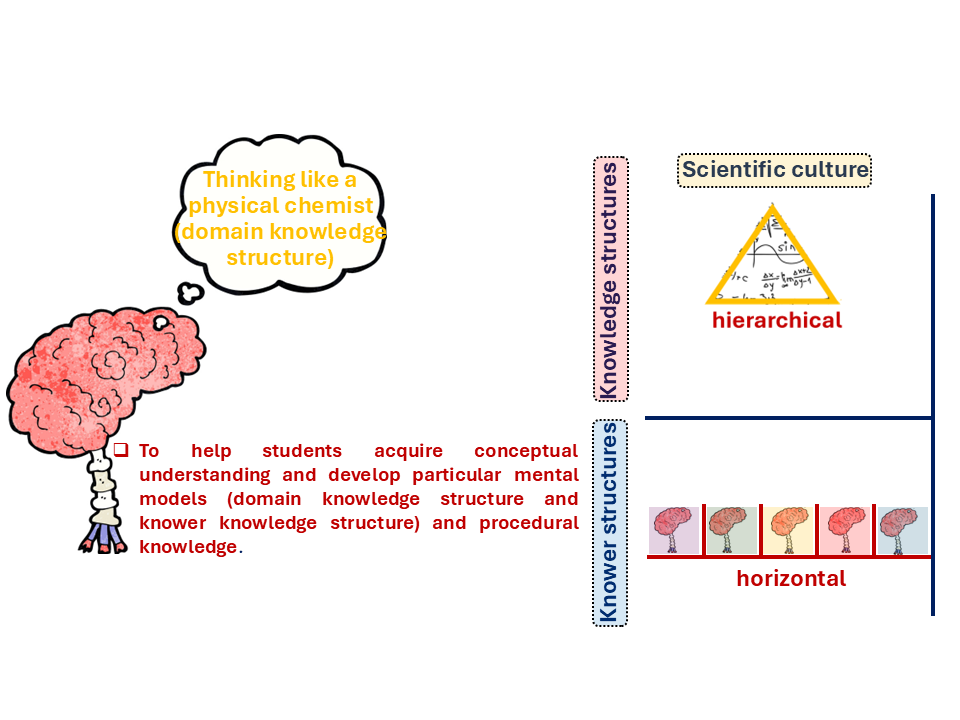
Figure 1: What Knowledge Looks Like in Physical Chemistry — Hierarchical knowledge structure (scientific culture, triangle) and horizontal knower structure.
Look at the two sides of Figure 1. On the right, Physical Chemistry sits in the scientific culture with a hierarchical knowledge structure (the triangle). Knowledge does not just accumulate here, it builds. In Physical Chemistry, concepts build on one another, understanding atomic structure is essential for grasping bonding, which in turn supports thermodynamic reasoning, all forming a layered framework that shapes explanations of chemical processes. As Bernstein (1999) put it, in disciplines like this, students only encounter the real depth of the subject very late in their education. Reaching that point requires every underlying layer to be in place first. That hierarchy is not arbitrary. It reflects how the knowledge is structured.
Now look at the knower structure beneath it, which is horizontal. The row of similar brains in Figure 1 makes the point clearly. In Physical Chemistry, unlike in music performance or fine art, there is no expectation that you bring a particular kind of self, a particular cultural background, or a special innate talent. Any student who masters the knowledge belongs. That is the discipline's genuine promise of openness, and it is real.
However, those three goals mentioned in Figure 1 (conceptual understanding, mental models, and procedural knowledge) do not develop together automatically. A student can build procedural knowledge without conceptual understanding. They can learn to execute without learning to think. And this is exactly where Physical Chemistry's promise runs into trouble. This happens because the hierarchy is demanding, and the examination system tends to reward the aspects that are easiest to assess (procedures), while deeper conceptual understanding can go unnoticed and undeveloped for years. The knowledge that earns a top grade and the knowledge that Physical Chemistry actually requires are not always the same thing. To understand why, we need to go one level deeper into those gold branches shown in Figure 1 of Blog 2.
What Those Gold Branches Actually Contain
Here is something that should unsettle any chemistry educator. Sadler and Tai (2007), studying over 18,000 students across 63 universities, found that high scores on pre-university science examinations correlate only modestly with performance in university science courses. Roughly half of even that modest advantage disappeared once they accounted for students' school backgrounds and access to resources. The grade was measuring something, but not the thing the university actually needed. A student can walk through the door with outstanding entry marks and still be profoundly unprepared for what Physical Chemistry demands.
That gap has a face. Park and Light (2009) followed three high-achieving students (all earning A grades ) through a college chemistry course on atomic structure. Only one had genuinely crossed into quantum thinking by the end. The third, Tom, could reproduce quantum numbers, describe orbital shapes, and invoke Heisenberg's uncertainty principle. He could perform. What he could not do was put any of it together into a coherent picture of what an atom actually is. His orbitals were still set paths for circling electrons, a Bohr model with quantum vocabulary painted over it. At one point, he noticed that in his own diagram, p-orbital electrons would run straight into the nucleus. He knew it was wrong. Then he moved on, because he had no framework to fix it.
Tom arrived with impressive entry marks. He left with a fundamentally broken mental model of the atom. The examination measured one kind of knowledge. The discipline required another. And the gap between them is exactly what the Epistemic Plane is designed to make visible.
Introducing the Epistemic Plane
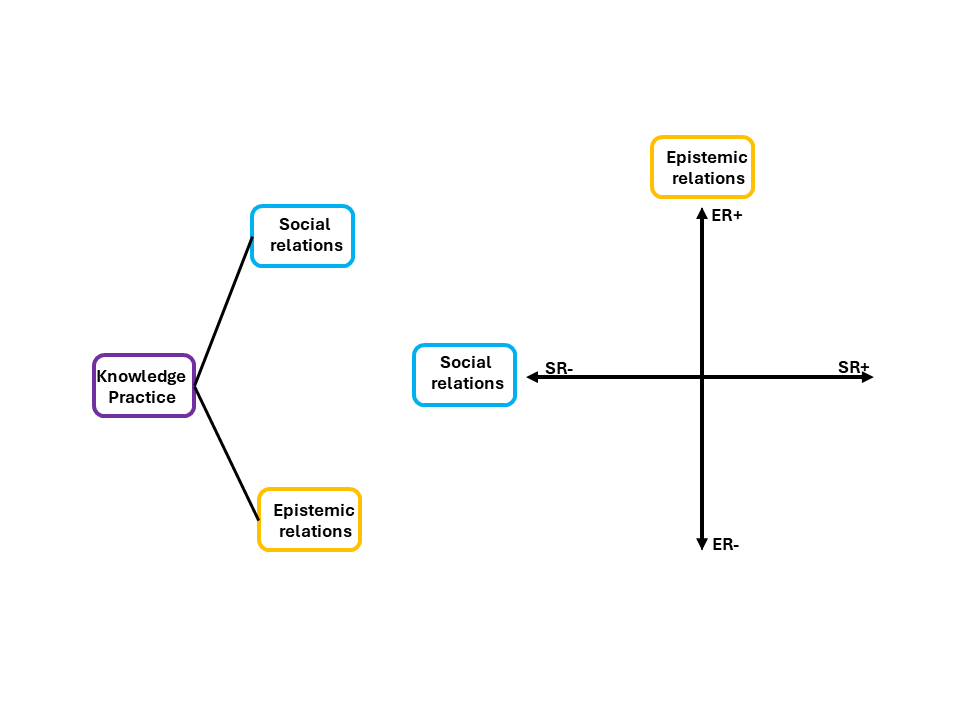
Before we go deeper, it helps to see the full picture first. Figure 2 shows how any knowledge practice, including Physical Chemistry, can be understood through two organising principles. On the left, you can see that a knowledge practice branches into two directions: social relations (the blue branch) and epistemic relations (the gold branch). On the right, those two branches become the axes of a plane. The vertical axis is epistemic relations, from weaker (ER−) at the bottom to stronger (ER+) at the top. The horizontal axis is social relations, from weaker (SR−) on the left to stronger (SR+) on the right. Where a discipline sits on that plane tells you what it values most.
Figure 2: Specialisation Plane adapted from Maton 2014.
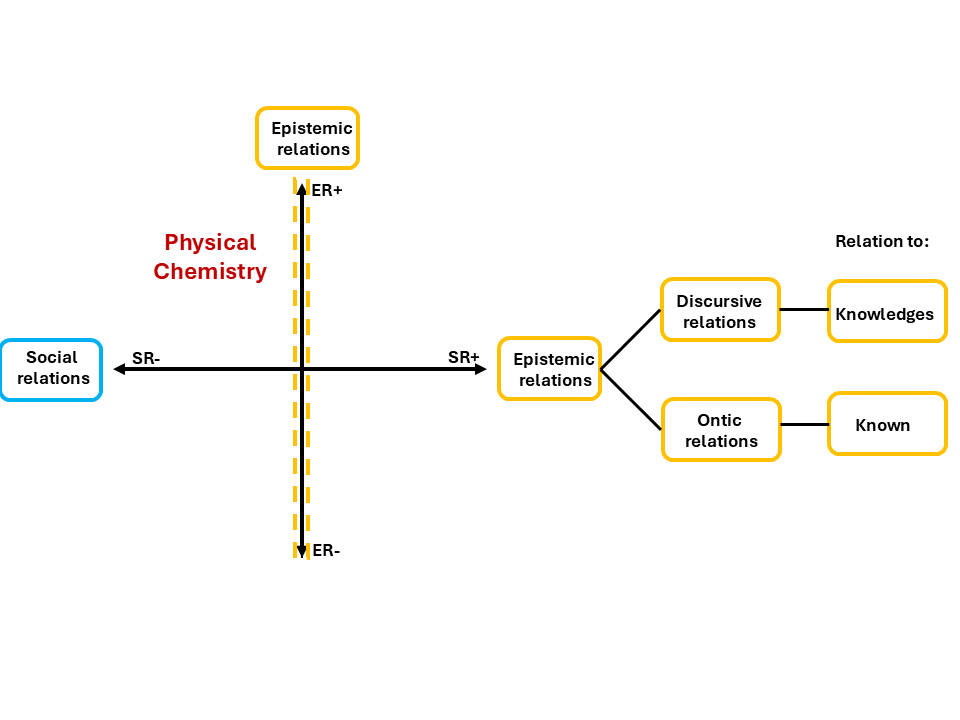
Physical Chemistry sits high on the vertical axis and to the left (strong epistemic relations, weaker social relations). What you know matters enormously. Who you are matters less. That is the knowledge code we established in the previous blog. This blog instead focuses on what sits inside that gold branch (epistemic relations), as this is where the difference between Tom's kind of knowledge and the kind Physical Chemistry actually requires becomes visible.
So what is actually inside those gold branches? As Figure 3 shows, epistemic relations are not one thing, but two. LCT breaks them open into two independent questions.
Figure 3: Specialisation plane focusing on the Epistemic relation, adapted from Maton(2014)
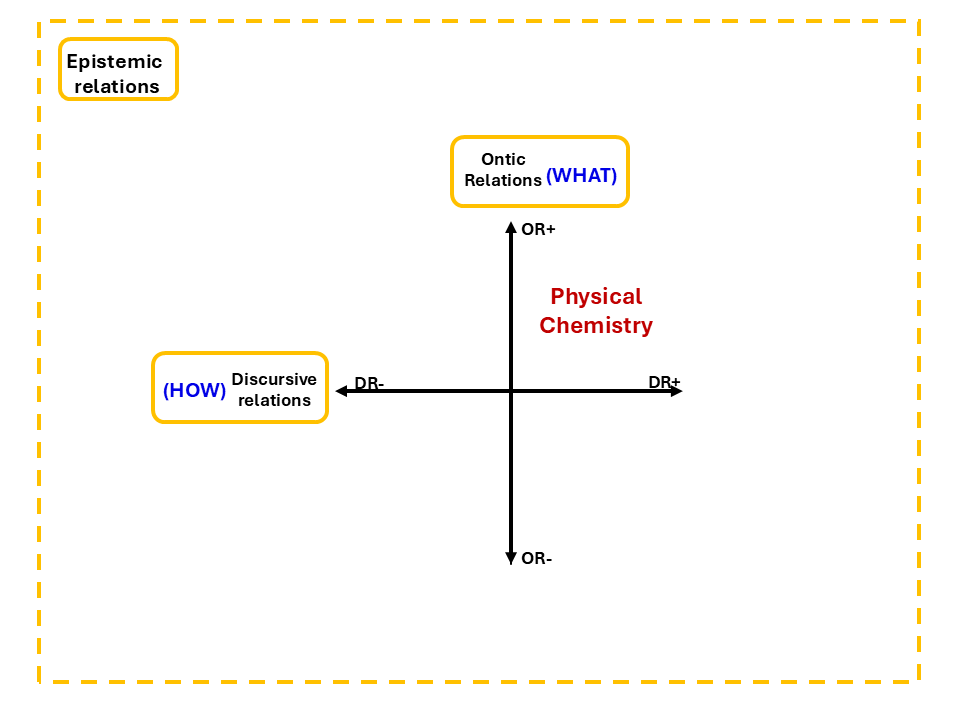
The first question is what the knowledge is about. LCT calls this ontic relations: how clearly defined is the object of study? Does the discipline know what it is investigating, or is its subject matter open and shifting? The second question is howthat knowledge is engaged with, the discursive relations: how standardised are the methods and procedures? Does the discipline insist on particular formalisms, or is there flexibility? These relations are summarised in Figure 4.
Figure 4: Epistemic Plane adapted from Maton (2014)
These two questions are independent of each other. Physical Chemistry sits firmly in the territory where both are strong. It is clear about what it studies. And it is equally clear about how those things are to be engaged, through mathematical formalisms, derivations, and standardised procedures that do not bend easily.
Four Ways of Knowing and Where Students Get Stuck
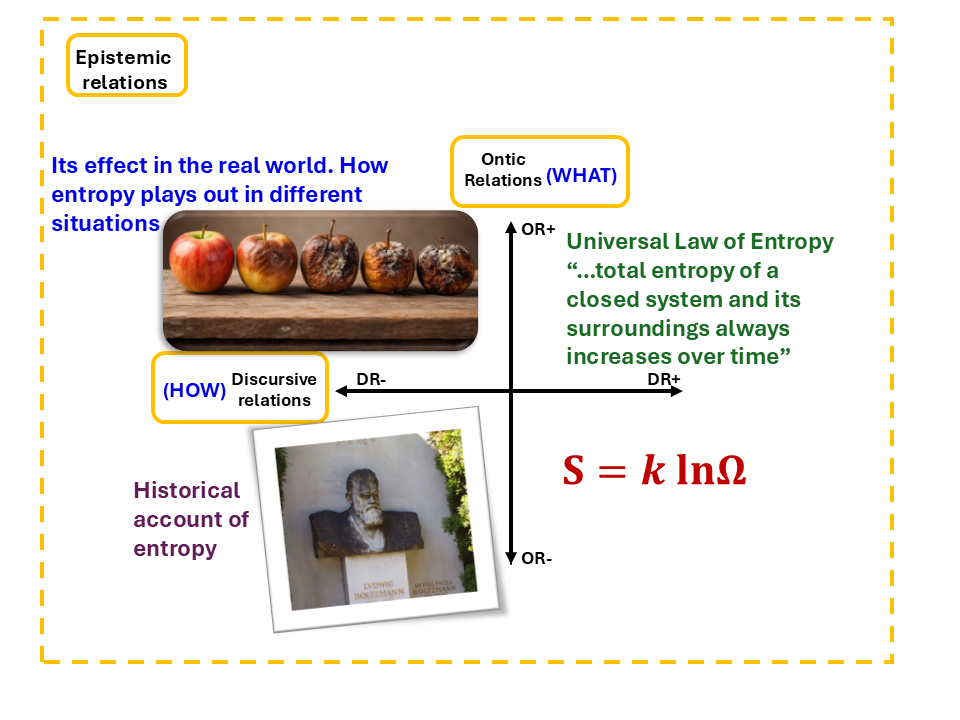
These two dimensions together create four different ways a student can be positioned in relation to the knowledge. Figure 5 uses entropy as an example to bring this to life, because entropy is arguably the concept where everything Physical Chemistry demands becomes most visible.
Figure 5: Epistemic relations
Ludwig boltzmann picture by Daderot at English Wikipedia - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=507663
Think about how differently students encounter it.
Some students meet entropy first as a story, Boltzmann's tragic biography, the intellectual battles of the nineteenth century, and the intuition that disorder always increases. It gives the concept warmth and human origin. On its own, however, this sits in what LCT calls knower insight: neither the object nor the method is tightly held. It does not constitute Physical Chemistry understanding. Quílez (2019) shows why this is a risky place to stay. Over time, entropy has been described using multiple metaphors (disorder, information, freedom, spreading, and heat), each highlighting different aspects, and there is no single consensus on the most precise interpretation. Students who stop here feel like they understand entropy. They have the story, but do not have the tools.
Other students can tell you what entropy does in the world. Heat flows from hot to cold. A perfume spreads across a room. Ice melts spontaneously at room temperature. Things turn to decay over time. This is situational insight: the object is clear (we are talking about entropy), but the approach is still open and context-dependent. This is real understanding. It is just not the understanding that Physical Chemistry assesses or builds on.
And then there is Tom's territory. Students who can calculate ΔS, apply the Gibbs equation, and determine spontaneity correctly (but who cannot connect any of it back to what entropy physically means) are in doctrinal insight. The procedure is right, however physical meaning has gone dark. Maton and Doran (2021) describe this as knowing the ingredients without knowing the recipe. Tom held quantum numbers, Heisenberg's principle, and orbital shapes as separate retrievable items. What was missing were the connections between them, the integrating idea of probability that would have made those pieces into a coherent quantum model. He could execute, but could not explain.
What Physical Chemistry actually requires is the upper-right quadrant, the purist insight. This means holding the object and the method together at the same time. For entropy, it means the Universal Law, the total entropy of a closed system and its surroundings always increases over time, held together with S=klnΩ in a way that makes the equation an act of physical reasoning, not just a formula to recall. Maton and Howard (2021) call this ontic condensation, the physical meaning becomes genuinely carried inside the formal representation. At the purist level, S=klnΩ is more than a formula, understanding it requires grasping the meaning of Ω, Boltzmann’s rationale for the logarithm, and the way this expression describes physical reality as an integrated whole. The formalism and the physical reality are not two separate things that happen to sit next to each other. They are fused, which makes it hard to reach, and harder still when nobody builds the pathway to get there.
Neumann, Parry, and Becher (2002) make an uncomfortable observation about disciplines like Physical Chemistry. They observed that students can often survive their courses with very little need to demonstrate this kind of integrated understanding. Assessment rewards what it can measure cleanly, the right formula, the reproduced derivation, and the correct calculation. That is doctrinal competence, it is valuable. It is not, however, purist insight, and passing the exam does not guarantee that deeper integration has taken place.
Claus et al. (2023), working in analytical chemistry, put the same point concretely. By making a point that a student can know every individual statistical parameter (standard deviation, limit of detection, calibration uncertainty) without ever crossing into understanding them as a unified framework that shapes what a chemist does in the laboratory. Before crossing that threshold, statistics is an exercise. After crossing it, statistics is a tool. The crossing requires explicit teaching, it does not happen through exposure to content alone.
The implication is important. Each step on the journey from knower insight to purist insight requires a deliberate bridge. Maton and Howard (2021) compared two science teachers and found that the one whose students genuinely integrated mathematical ideas into scientific understanding was the one who explicitly traced the pathway. The student who can separate the mathematical tool from the science, repurpose it for the scientific question, and then integrate it back into physical meaning. The other teacher left students with what one of them honestly described as "a lot of numbers", correct, and disconnected from the world they were meant to describe.
Concluding Remark
The Epistemic Plane asks a different question than the one most educators start with. Instead of why can't this student get it? it asks: where are they positioned, and what would it take to move them forward? That shift, from deficiency to positioning, changes what you design in a course, what you reward in an assessment, and how you think about a lecture.
Teaching that begins in abstraction and never leaves it produces doctrinal competence at best, students who can calculate but cannot explain. Teaching that stays only in real-world examples and never reaches the formalism leaves students without the tools to transfer their understanding to anything new. The journey across all four positions matters. And the bridges between them need to be designed deliberately, not left to chance.
Tom earned an A. He satisfied every measure the course put in front of him. And he left with a model of the atom he knew was wrong, because nobody built the bridge between his procedural knowledge and the physical understanding that would have made it coherent. He was not the problem, the teaching was. The Epistemic Plane gives us the language to say that clearly and, more importantly, the framework to design the teaching that might have actually helped him.
The Epistemic Plane, however, represents only one side of the picture. Recall from Figure 2, that the keys had two sets of branches (gold and blue). We have spent this entire blog on the gold. The blue branches (social relations) and what they say about who gets to belong in Physical Chemistry raise questions that are just as important, and just as uncomfortable. That is where we are going next.
Coming Next
The Social Plane: Who Gets to Belong in Physical Chemistry?
Physical Chemistry formally says that who you are does not matter, only what you know. The question, however, is whether this holds true in practice? The blue branches in Figure 1 (social relations) open up a different and more unsettling part of the picture. LCT's Social Plane asks who the discipline quietly expects its learners to be, and what happens to students whose backgrounds did not prepare them for expectations that were never made visible. That is the focus of the next blog.
References
Bernstein, B. (1999). Vertical and horizontal discourse: An essay. British Journal of Sociology of Education, 20(2), 157–173.
Claus, M.J., Gionfriddo, E., Monahan, J., Whelan, R.J. & Vitha, M.F. (2023). Threshold concepts in analytical chemistry. Journal of Chemical Education, 100, 427–431.
Maton, K. (2014) Knowledge and knowers: Towards a realist sociology of education. London: Routledge.
Maton, K. and Doran, Y. J. (2021) ‘Constellating science: How relations among ideas help build knowledge’, in Maton, K., Martin, J. R. and Doran, Y. J. (eds.) Teaching science: Knowledge, language, pedagogy. London: Routledge, pp. 49–75.
Maton, K. and Howard, S. K. (2021) ‘Targeting science: Successfully integrating mathematics into science teaching’, in Maton, K., Martin, J. R. and Doran, Y. J. (eds.) Teaching science: Knowledge, language, pedagogy. London: Routledge, pp. 23–48.
Neumann, R., Parry, S. and Becher, T. (2002) ‘Teaching and learning in their disciplinary contexts: A conceptual analysis’, Studies in Higher Education, 27(4), pp. 405–417.
Park, E. J. and Light, G. (2009) ‘Identifying atomic structure as a threshold concept: Student mental models and troublesomeness’, International Journal of Science Education, 31(2), pp. 233–258.
Quílez, J. (2019) ‘A categorisation of the terminological sources of student difficulties when learning chemistry’, Studies in Science Education, 55(2), pp. 121–167.
Sadler, P. M. and Tai, R. H. (2007) ‘Advanced Placement exam scores as a predictor of performance in introductory college biology, chemistry and physics courses’, Science Educator, 16(2), pp. 1–19.